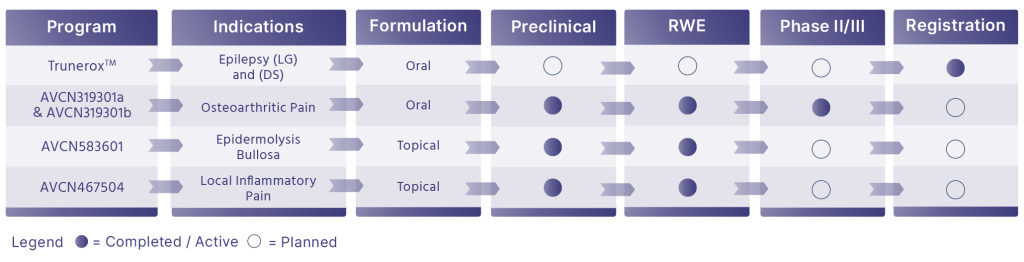
Pharmaceutical Pipeline of Indication-Specific Cannabinoid Therapeutics
Leveraging an Integrated Platform to Develop Proprietary, Evidence-Based Drug Candidates
Trunerox™ 10% CBD Oral Solution
Adjunctive Treatment Of Seizures Associated with Lennox Gastaut Syndrome (LGS) and Dravet Syndrome (DS)
Trunerox™ is a pharmaceutical drug with proprietary formulation manufactured under GMP standards and utilizing the Company’s purified API. Clinical indications show Trunerox™ shows potential as an adjunctive treatment for seizures associated with Lennox Gastaut Syndrome (LGS) and Dravet Syndrome (DS).
Marketing authorization has been obtained in Colombia by INVIMA in Q1, 2024. Commercialization in Colombia took place in late 2025.
Pharmaceutical Pipeline
Indication-Specific Cannabinoid Therapeutics
Leveraging an integrated platform to develop proprietary, evidence-based drug candidates.